Company Size
+350 employees

Traphaco 3S ERP Digital Transformation
Solutions
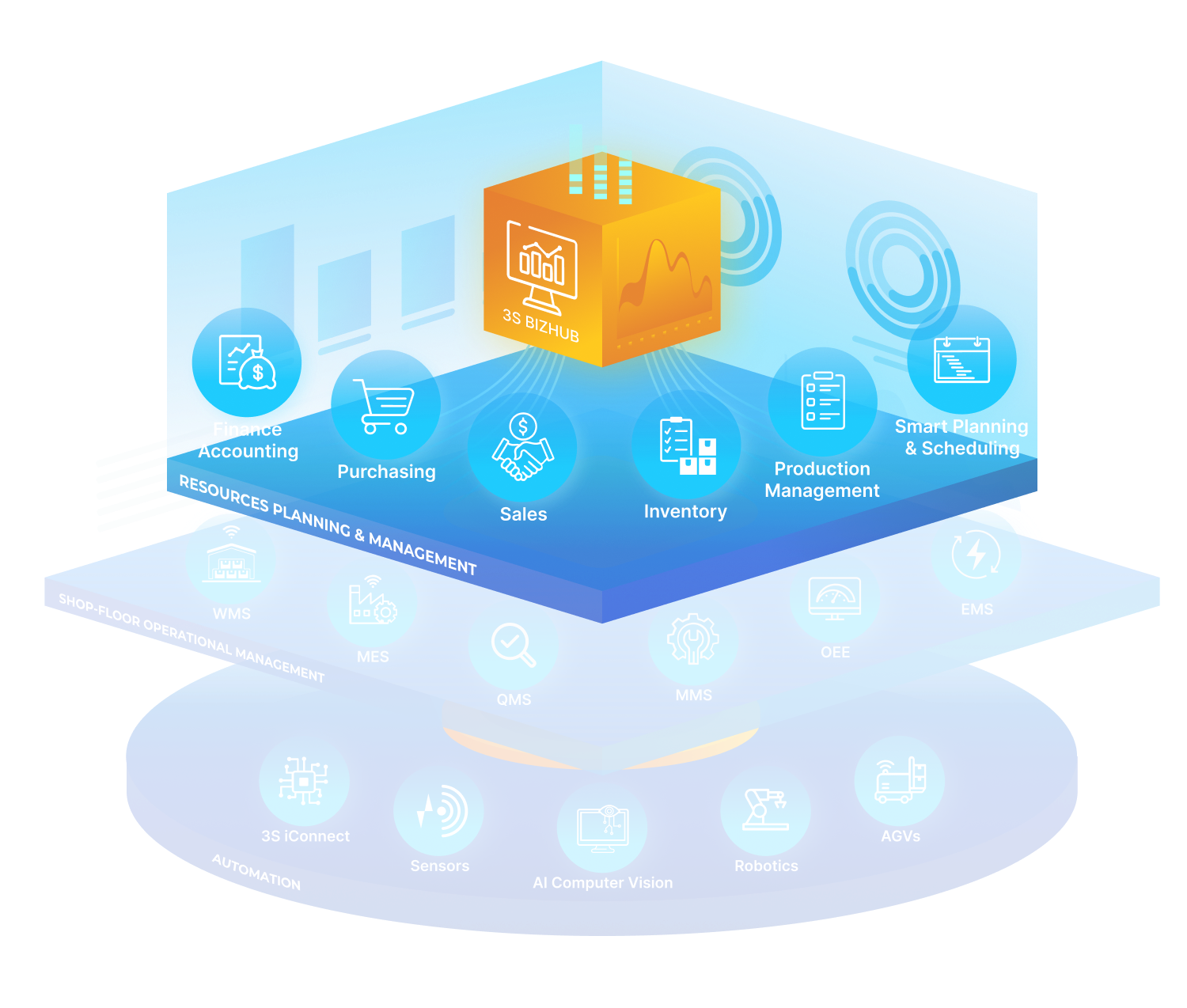
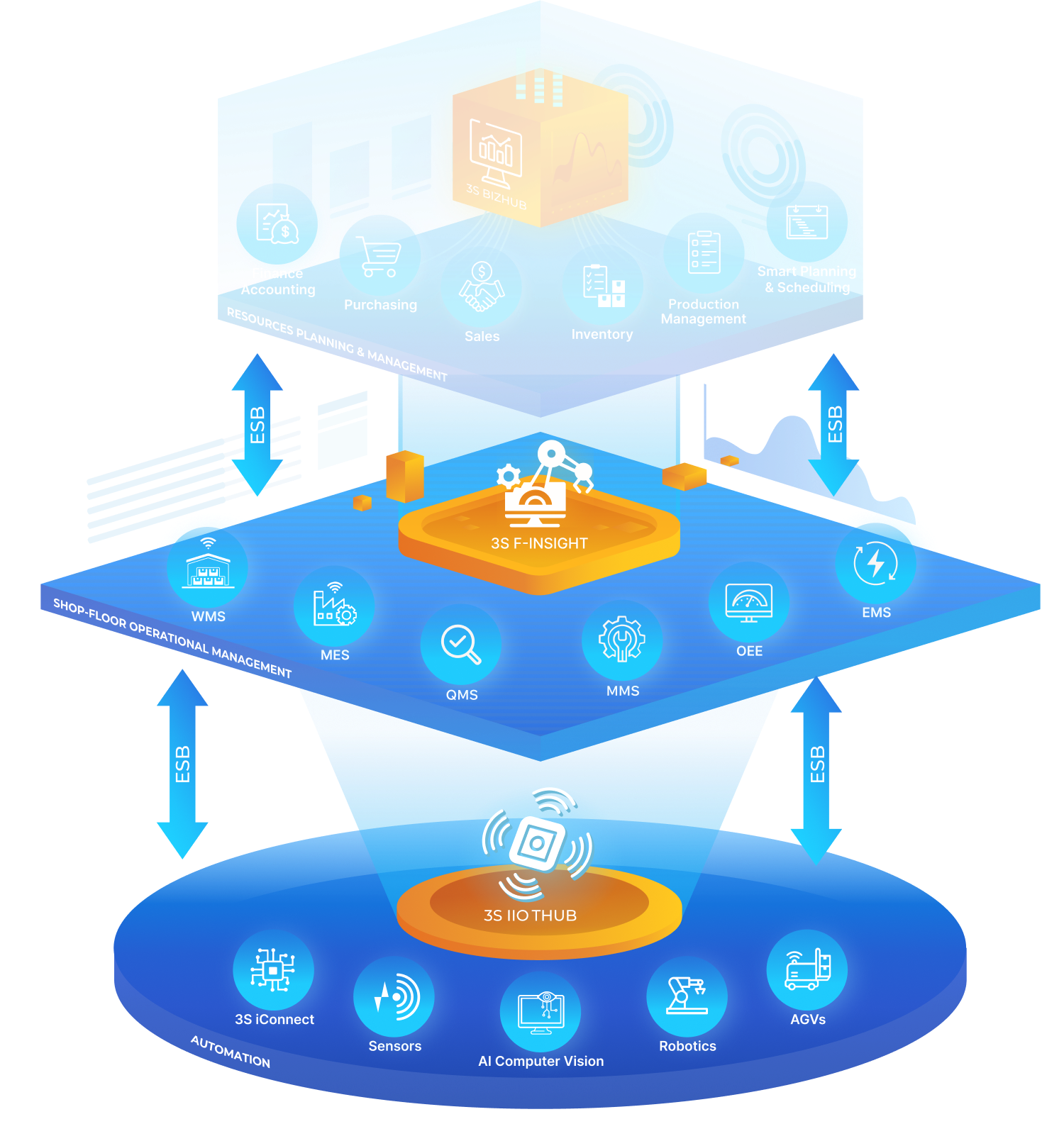
3S ERP
Results
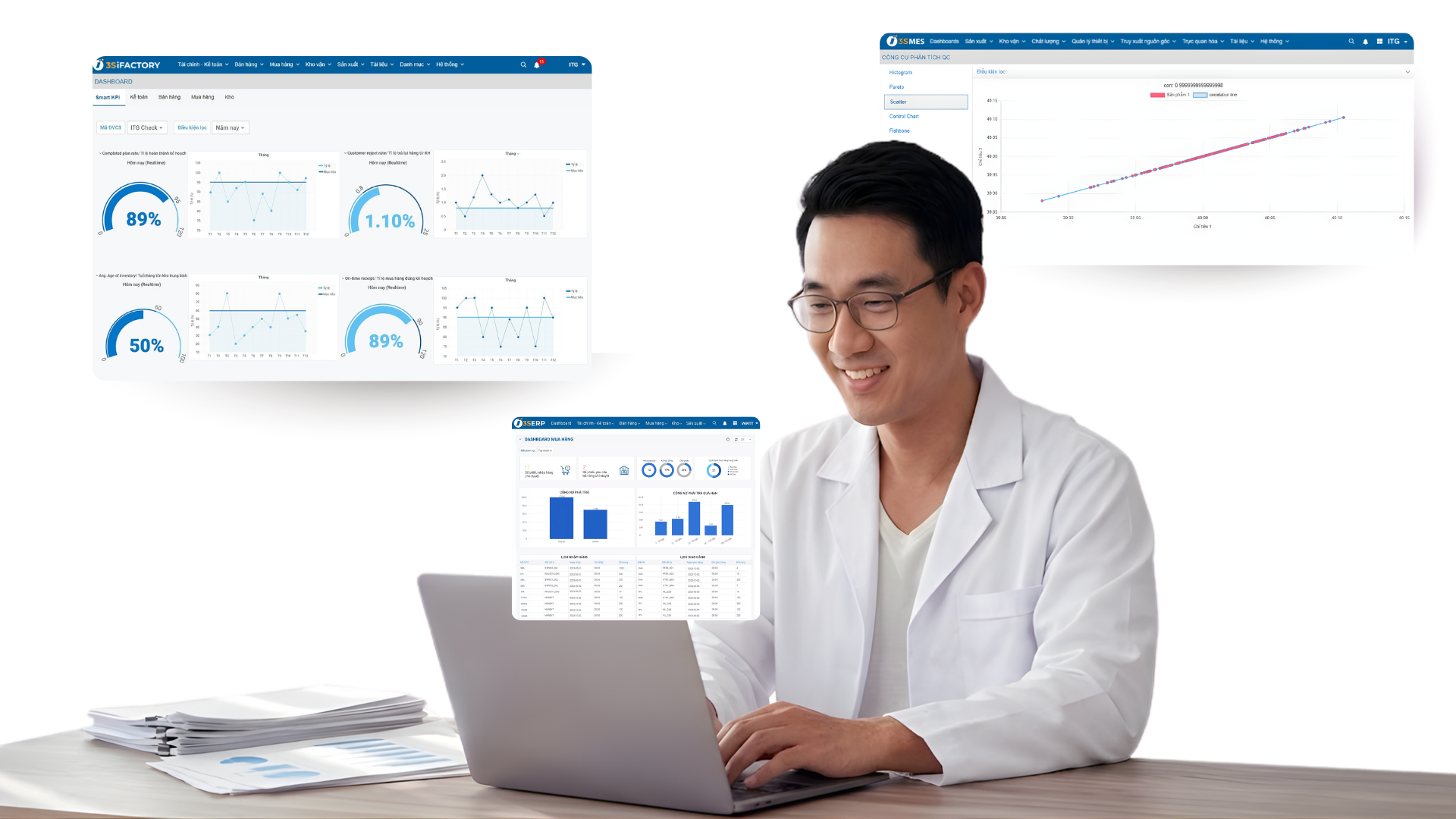
- Comprehensive Digitization: 100% of data and Batch Records migrated to a digital environment.
- Quality Governance: Strict process control from raw materials to finished products (GMP standards).
- Operational Optimization: 100% reduction in paper storage costs with instantaneous origin traceability.
- Intelligent Administration: Real-time visual reporting and monitoring via mobile devices.